Introduction
Plastics and composites are of course non-metallic materials, but they tend to be considered as their own special groups rather than being considered within the larger group of non-metallic materials. Rather, non-metallic materials tends to consider some extremely common materials that are so ubiquituous that we often simply do not think about them. Nevertheless, they are still engineering materials – possibly the most common and longest used of all.
Over the course of this section we will look at them, and by the end you will have developed an understanding of the properties and applications of:
- Engineering ceramics
- Glasses
- Rock and stone
- Wood
Ceramics and glasses
Ceramics can be split into two major groups:
- Engineering ceramics which are used for cutting tools, engine parts and wear-resistant parts amongst others.
- Vitreous ceramics – clay products used in crockery, tiles etc.

Figure 1 Vitreous ceramics exist in a wide array of designs and colours. [Pixabay]
Glasses come in a wide array of types with a broad array of properties, however they are all based on silica, the giant lattice form of silicon dioxide SiO2.
Some of the advantages and disadvantages of ceramics are discussed in the next two images.
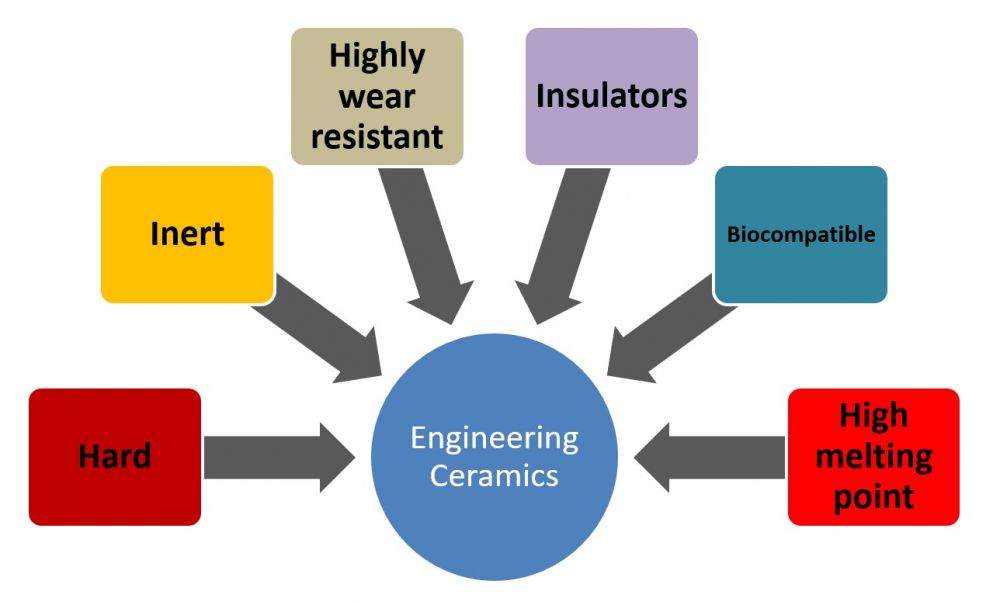
Figure 2 Advantages of engineering ceramics
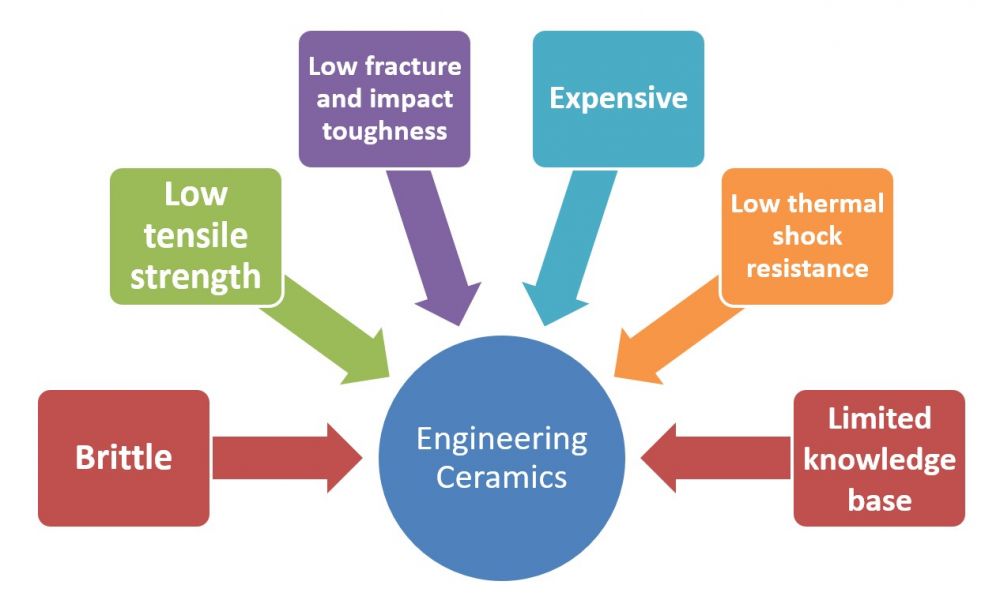
Figure 3 Disadvantages of engineering ceramics
The atomic structure of ceramics depends on the elements from which they are made, and can be either ionic or covalent.
Ionic ceramics are compounds of a metal and a non-metal, for example:
- Magnesium oxide (MgO)
- Alumina (Al2O3)
- Zirconia (ZrO2)
Covalent ceramics on the other hand are compounds of two non – metals, or can be a pure element, for example:
- Diamond (C)
- Silicon carbide (SiC)
- Silicon nitride (Si3N4)
Engineering ceramics are generally manufactured from powders that are compacted and then sintered. The sintering of fine powders will occur at temperatures as low as 2/3Tm (melting point of the powder). The finer the powder, the smaller the resultant pores and the better the mechanical strength. The fusing of the particles typically results in a grain size larger than the powder. Higher densities and smaller grain sizes can be obtained by applying pressure and heat at the same time.
Looking at the microstructure, Ceramics (but not glasses) form polycrystalline structures in a comparable manner to metals, with grains meeting each other at grain boundaries. Most ceramics contain porosity, up to 20%, and these weaken the structure, although the affect of this porosity on the strength is less than those caused by microcracks.
Microcracks (small cracks) are also present in most ceramics, the result of processing or thermal or mechanical stresses. These have a much greater influence on the strength. The toughness of ceramics can be improved by the addition of fibres, resulting in a ceramic matrix composite material.
Glasses have similar properties to ceramics in that they hard, inert, brittle, etc. One way in which they differ is that they have an amorphous (an irregular non-crystalline) microstructure.
Most glasses are pure silica (silicon dioxide, SiO2), based, but can also contain other compounds. They are often transparent.
Some of their properties, such as toughness and thermal shock resistance, can be enhanced through thermal or chemical processes. They are commonly used for glazing, drinks containers and a host of other items, and they can also be drawn into fibres for use in composite materials.
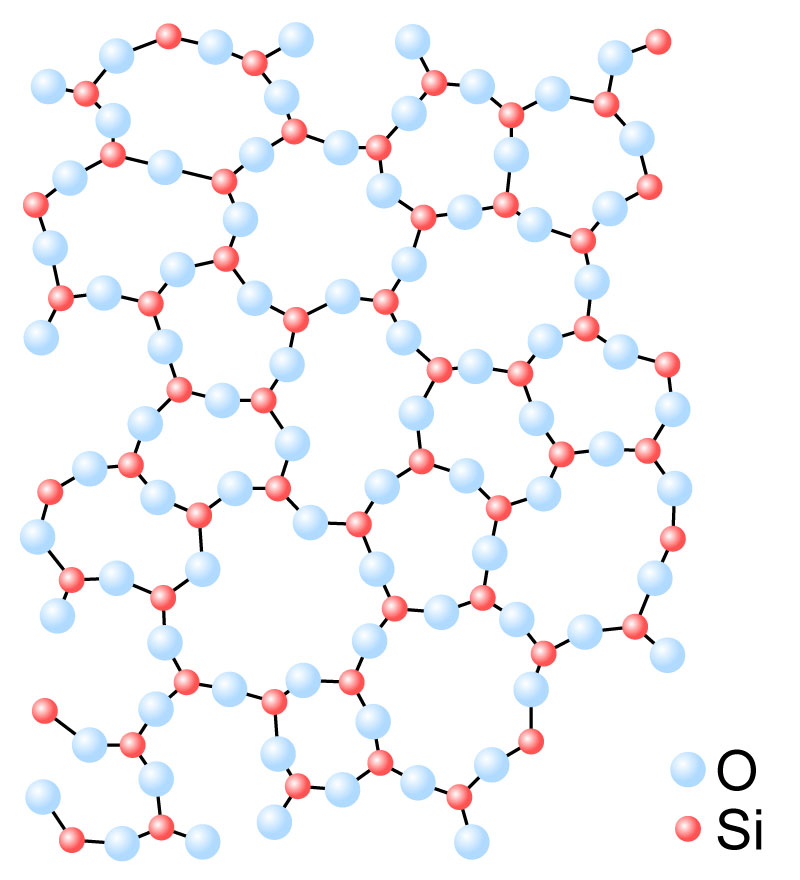
Figure 4 Graphical representation of the structure of silica. [Wikimedia]
Rock/Stone
Rocks/stones have comparable properties to ceramics in that they are often hard, brittle and have high melting points. The main difference in terms of classification is that rock and stone is naturally occurring, ceramics are usually man made.
They typically have a granular or crystalline microstructure, the exact microstructure depending on the specific type of rock and a host of other factors. Rocks/stones have been used by humans for thousands of years with uses ranging from axe heads to the construction of roads and buildings, and remain one of the most commonly used engineering materials worldwide.

Figure 5 Red sandstone as seen in this picture of the Scottish town of Kirriemuir is a soft material that wears is easily, and yet has been used extensively as a building material, primarily due to its ease of working and proximity, but also for its pleasant colour. [Wikimedia / CC BY-SA 2.0]
Rock and stone are usually classified as belonging to one of three groups:
- Igneous – formed through the solidification of magma or lava. Granite and pumice are examples.
- Sedimentary – formed at the earth’s surface through the accumulation of sediments. Shale, limestone and sandstone are examples.
- Metamorphic – formed through the application of heat and pressure to either igneous or sedimentary rocks (or a previous metamorphic rock) resulting in a change in its properties. Slate and marble are examples.
.jpg?1641919051388)
Figure 7 Layers of sedimentary rock in Maktesh Ramon. [Wikimedia / CC BY-SA 4.0]

Figure 8 Metamorphic rock along Georgian Bay, Ontario, Canada. [Wikimedia / CC BY-SA 4.0]
Wood
Woods differ from metals and all the other materials that we have looked at so far in that they are organic materials. They are heterogeneous (their microstructure consists of two or more distinct phases) and anisotropic (their properties vary depending in which direction they are measured). Heterogeneity and anisotropy are also properties of composite materials.
Unlike the other materials we have examined, woods can be affected by changes in climatic conditions, resulting in a change in size, shape and strength. In addition it does not suffer from corrosion, but is susceptible to attack by fungi and insects.
Woods are usually grouped into soft and hard woods.
They are possibly the longest used of all engineering materials, having been used by man for thousands of years. Interestingly, structural wood reacts surprisingly well in fires, becoming stronger with heat – until it catches fire of course! This is unlike steel and other structural materials which get weaker as they heat up.

Figure 9 Whilst this "log" was once wood, it has petrified over many years to become stone in the Petrified National Park, Arizona, USA. [Wikimedia]
Soft Woods
Softwoods are derived from evergreen trees such as pine that are usually quicker growing than hardwoods. They are often softer and easier to work than hardwoods, have a high strength to weight ratio and are usually less expensive than hardwoods.
Common types are sikta spruce, Douglas fir, white pine, and they have many applications including construction of houses including joists, framework, roof trusses, furniture, boat building.
Hard Woods
Hardwoods are derived from both evergreen and deciduous trees – though most commonly from deciduous trees. Hardwoods are usually more dense and stronger than softwoods, although balsa is a (soft) hardwood and yew is a (hard) softwood.
Common types are mahogany, birch, oak and ash. As with softwoods, they have many applications such as furniture, musical instruments, flooring. They are generally more expensive than softwoods as they are slower growing, so they tend to be used in higher specification situations.
Interestingly, some hardwoods – such as greenheart (Chlorocardium rodiei) – are so dense they do not float. Greenheart was used in the production of many polar exploration ships such as Scott’s Discovery and Shackleton’s Endurance as it was so strong it would prevent the ships being crushed should they be surrounded by ice.
Technically, wood is a composite material consisting of hollow pipes made from cellulose and hemicellulose. These are surrounded, and held together, by lignin (acting as the matrix).
If we look at the microstructure we will see that hardwoods contain pores for carrying water (upper image) whereas softwoods (lower image) do not.

Figure 10 Microstructure of Hard and Softwoods. [Wikimedia]
Given the variability between different types of wood, and even within wood of the same type due to atmospheric and environmental effects, it is not possible to directly attribute properties to wood as a whole, but it is possible to define certain characteristics. Such as:
- High strength to weight ratio.
- High modulus of elasticity, albeit depending on the direction of the grain. The elastic modulus typically ranges from 1 – 20 GPa, in comparison aluminium is 70 GPa and steel 205 GPa.
- Strong in tension, compression and bending, although this is dependent on the grain direction.
- Easily formed bonded joints.
- Easy to cut and shape.
- Low cost.
- Relatively easy to repair.
The tensile strength of wood is high in the grain direction, but low across it. For example, spruce has a tensile strength of ~100 MPa in the grain direction, although only ~3 MPa across the grain.
The compressive strength is what usually limits the load that can be taken by a member. Excessive compressive strength results in compression creases (buckling) of the tubular structure and permanent damage.
The shear strength is the ability of the wood to resist internal slippage along the grain. In wood the typical shear strength is less than one tenth that of the tensile strength.
The graph shows where various materials – including woods – lie in terms of tensile strength against density.

Graph 1 Density vs Tensile strength of various materials. Drawn by Gareth Bradley, used with his permission.
Engineering woods take the natural wood and apply various processes to them to change their properties. They consist of small particles or layers of wood glued together. Examples are plywood, chipboard, medium density fibreboard, and hardboard.















